| Journal of Clinical Gynecology and Obstetrics, ISSN 1927-1271 print, 1927-128X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Gynecol Obstet and Elmer Press Inc |
| Journal website https://jcgo.elmerpub.com |
Short Communication
Volume 000, Number 000, March 2025, pages 000-000
Intravenous Estrogen for Acute Heavy Menstrual Bleeding: Commentary and Database Analysis
Amanda V. Frencha, e, Nancy Sokkaryb, Bethany Samuelson Bannowc, Divyaswathi Citla-Sridhard, e
aDepartment of Obstetrics and Gynecology, Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA
bDepartment of Obstetrics and Gynecology, Children’s Healthcare of Atlanta, Emory University School of Medicine, Atlanta, GA, USA
cHemostasis and Thrombosis Center at OHSU, Oregon Health & Science University, Portland, OR, USA
dDepartment of Pediatric Hematology and Oncology, Arkansas Children’s Hospital, University of Arkansas for Medical Sciences, Little Rock, AR, USA
eCorresponding Author: Divyaswathi Citla-Sridhar, Department of Pediatric Hematology and Oncology, Arkansas Children’s Hospital, University of Arkansas for Medical Sciences, Little Rock, Arkansas, USA; Amanda V. French, Department of Pediatric Surgery, Massachusetts General Hospital, Boston, MA 02114, USA
Manuscript submitted December 17, 2024, accepted March 19, 2025, published online March 25, 2025
Short title: Intravenous Estrogen and VTE
doi: https://doi.org/10.14740/jcgo1025
| Abstract | ▴Top |
Background: Heavy menstrual bleeding (HMB) is common. Although hormonal medication is the mainstay of treatment, there is no scientific evidence to support the superiority of one regimen over another. Both oral and intravenous (IV) forms of estrogen are used for HMB in the acute setting and are known to be associated with thrombosis, in particular, venous thromboembolism (VTE) in the deep veins of the legs or pulmonary vessels. Progestins are also used for treatment of HMB; a common choice is norethindrone acetate, a small amount of which is metabolically converted to ethinyl estradiol (an estrogen) after ingestion. We sought to assess the incidence and relative risk (RR) of VTE for users of IV estrogen, oral ethinyl estradiol, and norethindrone acetate.
Methods and Results: A retrospective descriptive review of a large de-identified database (TriNETX, LLC) revealed a significantly higher calculated rate and RR of VTE amongst users of injectable conjugated estrogen compared to users of oral ethinyl estradiol and oral norethindrone acetate.
Conclusion: Research is needed to determine the true RR of thrombosis for IV estrogen users to allow clinicians and patients to make informed decisions that appropriately stratify risks and benefits when considering the options for hormonal treatment of acute HMB.
Keywords: Intravenous estrogen; Conjugated estrogen; Thrombosis; Venous thromboembolism; Heavy menstrual bleeding
| Introduction | ▴Top |
Heavy menstrual bleeding (HMB) is defined as blood loss exceeding 80 mL during one menstrual cycle, and is accompanied by clinical signs including menses lasting longer than 7 days, a need to change protection more often than every 1 - 2 h and iron deficiency, with or without anemia [1]. This type of bleeding can be described with the 2018 International Federation of Gynecology and Obstetrics (FIGO) abnormal uterine bleeding (AUB) system [2, 3]. HMB affects approximately one-third of menstruating people. Anemia may be severe (hemoglobin < 8 g/dL) and accompanied by hypovolemia and hemodynamic instability requiring acute inpatient management. Acute HMB, therefore, is AUB/HMB requiring immediate medical attention. Hormonal medication is typically used to stabilize the endometrium and stop or significantly diminish active bleeding [4]. Intravenous (IV) estrogen has long been incorporated into clinical treatment algorithms, and is often considered first line for severe acute HMB, particularly for individuals unable to tolerate oral medication [5]. There is no evidence to support the superiority of one hormonal medication regimen over another for AUB/HMB treatment; choices include varying formulations and routes of administration for progestin, estrogen, or a combination of both.
In a 1982 double-blind randomized controlled trial (RCT) of 34 patients (16 - 49 years) with dysfunctional uterine bleeding, 18 received 25 mg IV conjugated equine estrogen, and 16 received a placebo. According to protocol, participants who continued to bleed received a second dose of study drug at 3 h and an open-label dose of estrogen at 5 h for a maximum total of three injections. After one injection, bleeding stopped in 4/18 (estrogen) versus 5/16 (placebo) (P = 0.226, odds ratio (OR) 0.71). After two injections, 9/18 (estrogen) versus 1/16 (placebo) stopped bleeding (P = 0.015, OR 11.57). By 5 h, 5/18 (estrogen) and 8/16 (placebo) were still bleeding [6]. Authors of this study concluded that IV conjugated estrogen was more effective at cessation of bleeding when compared to placebo, and opined that the IV route may act more quickly than the oral route, based on their data showing that 72% of the estrogen arm participants stopped bleeding in under 3 h. Citing this study, the American College of Obstetrics and Gynecology (ACOG) Committee Opinion #557 currently recommends, “in the absence of contraindications to estrogen, hormonal therapy for acute HMB can consist of intravenous conjugated estrogen (25 mg) every 4 - 6 h (for 24 h); alternatively, monophasic combined oral contraceptive pills (OCPs), in 30 - 50 µg ethinyl estradiol formulation, can be used every 6 - 8 h until cessation of bleeding” [4]. This Committee Opinion further states that “studies of treatment of acute AUB are limited, and only one treatment (IV conjugated equine estrogen) is specifically approved by the United States Food and Drug Administration for the treatment of acute AUB.”
However, estrogen is known to increase thrombosis risk, in particular, venous thromboembolism (VTE). Progestins are generally considered safer for individuals at risk for thrombosis, as they are associated with a lower risk of VTE, though there is some variation depending on progestin type. Norethindrone acetate, when used for treatment of HMB, and depot medroxyprogesterone have been associated with increased odds of thrombosis when compared to controls as well as to other progestins, though this risk is still lower than that associated with ethinyl estradiol, which is a common component of combined OCPs [7]. Combined OCPs with third and fourth generation progestins (desogestrel, gestodene, chlormadinone acetate, cyproterone acetate, dienogest, and drospirenone) have been associated with a higher risk of VTE than the first and second generation alternatives (lynestrenol, norethindrone, levonorgestrel, norgestimate, and norgestrel) [8]. Elevated or exogenous estrogen has also been associated with unusual site thrombosis and arterial thrombosis [5]. Although the exact pathophysiology is not clear, estrogen is known to affect hemostatic pathways in multiple ways, including increasing levels of fibrinogen, factors II, VII, VIII, and X [8], affecting platelet aggregation and activation, and elevating von Willebrand factor levels [9]. Most studies have shown that higher doses of estrogen are associated with higher VTE risk [10-13] and risk of thrombosis is highest in the first 6 months of oral estrogen use [5]. IV estrogen administration bypasses the liver, resulting in higher bioavailability compared to the oral route, but IV preparations are generally reserved for short-term treatment, and many patients may be given only a single dose. With the aim of adding to these recommendations, we sought to assess the incidence and relative risk (RR) of VTE for users of IV estrogen, oral ethinyl estradiol, and oral norethindrone acetate using a large, administrative database.
| Materials and Methods | ▴Top |
This was a retrospective descriptive study. Population level data were collected from TriNETX, LLC, an administrative database that contains inpatient and outpatient data from 56 tertiary care hospitals in the United States and > 81 million patients. TriNetX aggregates real-world de-identified data from patient electronic medical records (EMRs) and includes medical diagnosis codes and medications. As this is a de-identified dataset with access to multiple institutions, this study was exempt from Institutional Review Board approval for all participating institutions. The study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
This study analyzed the prevalence of VTE among females aged 10 - 45 years using the TriNetX database from January 1, 2000, to December 30, 2023. Patients were categorized into three age-based groups: group 1 (adolescents and young adults, 10 - 21 years), group 2 (reproductive age, 22 - 34 years), and group 3 (peri-menopausal, 35 - 45 years). These age groups were selected based on hormonal use patterns, physiological differences between the groups, and VTE risk stratification, aligning with established clinical guidelines and the US Medical Eligibility Criteria (US MEC) for contraceptive use. Patients were further classified into four hormonal therapy exposure groups: no hormonal therapy (control group), oral norethindrone acetate, oral ethinyl estradiol, and injectable conjugated estrogen. Hormone exposure was identified using RxNorm codes, and VTE events were defined as the presence of ≥ 1 ICD-9 or ICD-10 codes for VTE diagnosis and a concurrent anticoagulant prescription within 1 year of hormone exposure. A detailed list of anticoagulants, including antiplatelets, injectable agents, vitamin K antagonists, and direct oral anticoagulants, is provided in Supplementary Material 1 (jcgo.elmerpub.com).
Norethindrone acetate, commonly used for HMB, is available in 5 mg oral tablets and is metabolized to norethindrone after ingestion. Since norethindrone (0.35 mg) is not typically used for acute HMB treatment but shares the same RxNorm code as norethindrone acetate, the 0.35 mg dose was excluded, with the assumption that the remaining data represented oral norethindrone acetate. Oral ethinyl estradiol was selected as it is the most common estrogen component of combined hormonal contraceptives in the USA. For conjugated estrogen, the database does not differentiate IV administration, so the term “injectable” was used as a surrogate.
Statistical analysis
Categorical variables, including age groups, hormone exposure categories, and VTE occurrence, were reported as frequencies and percentages. Prevalence rates of VTE were calculated for each hormone exposure group (oral norethindrone, oral ethinyl estradiol, and injectable conjugated estrogen) and compared using Chi-square or Fisher’s exact test, as appropriate. Age-stratified analyses were performed by categorizing patients into three groups: 10 - 21 years, 22 - 34 years, and 35 - 45 years. Within each age group, VTE rates were compared across hormone exposure categories to assess potential differences in risk. Due to low sample size, injectable conjugated estrogen use in the 10 - 21 age group was excluded from statistical comparisons. Statistical significance was set at P < 0.05. All statistical analyses were conducted using MedCalc 18.2.1 (MedCalc®).
| Results | ▴Top |
The analysis included 25,791,533 females aged 10 - 45 years from the TriNetX database. Among these, the subset without active hormonal medication use showed a rising trend in the incidence of VTE between 2010 (0.10%) and 2020 (0.27%), indicating the baseline prevalence of VTE independent of hormonal exposure. Collected data were then stratified by age and each group was analyzed separately (Fig. 1).
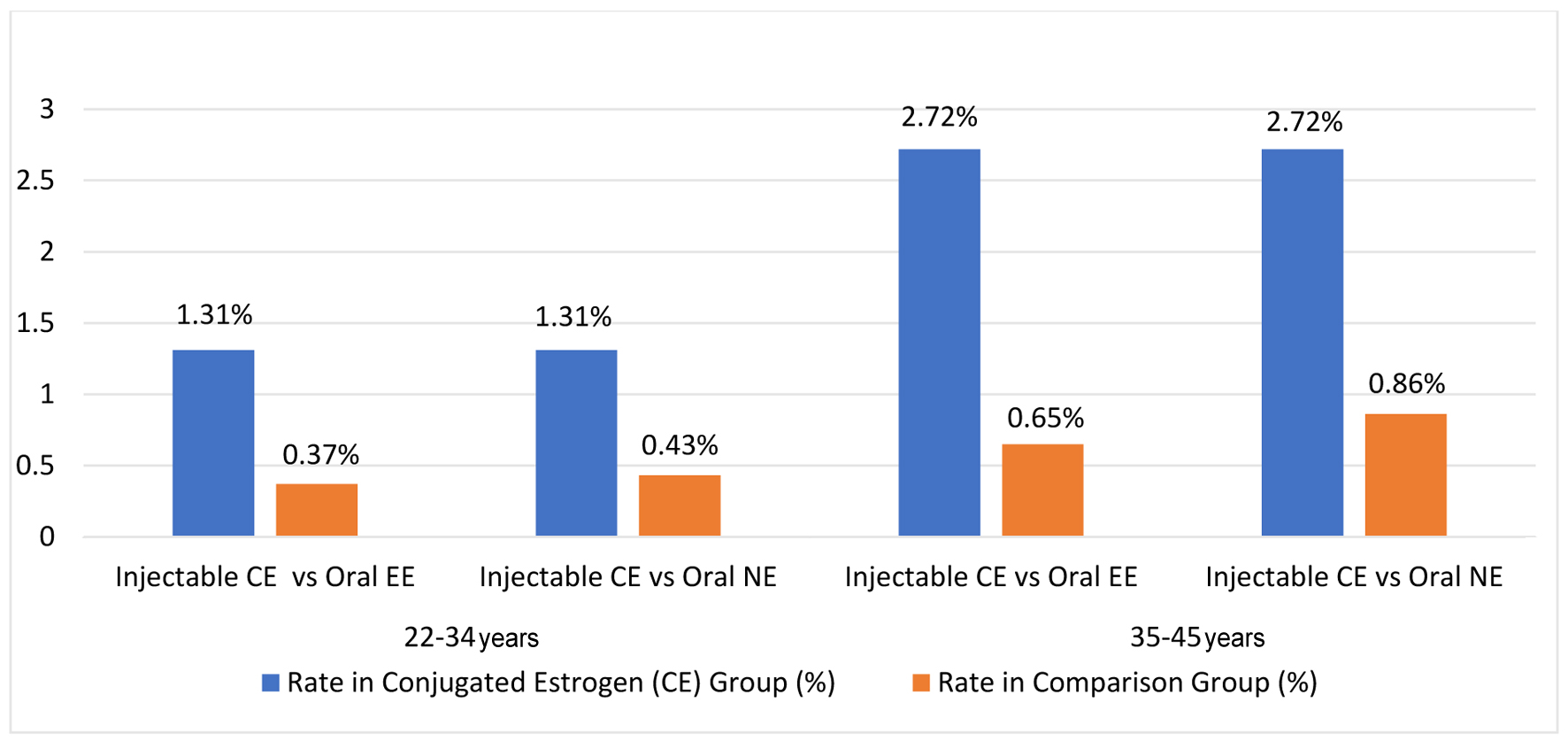 Click for large image | Figure 1. Comparison of rates of VTE by hormone type and age. CE: conjugated estrogen; EE: ethinyl estradiol; NE: norethindrone; VTE: venous thromboembolism. |
Age group 10 - 21 years
In younger females, oral norethindrone users (n = 75,387) demonstrated a VTE rate of 0.31% (there were 236 VTE events), which was more than double the rate observed in oral ethinyl estradiol users (n = 184,189, VTE rate: 0.14%, 250 VTE events). Due to low numbers, injectable conjugated estrogen use in this age group could not be meaningfully analyzed.
Age group 22 - 34 years
For females aged 22 - 34, VTE rates varied significantly by hormone type and administration route. Oral ethinyl estradiol users (n = 921,631) experienced a VTE rate of 0.37% (3,383 VTE events), while norethindrone users (n = 384,423) had a slightly higher rate of 0.43% (1,658 VTE events). Injectable conjugated estrogen users (n = 990) had a markedly higher VTE rate of 1.31% (13 VTE events), representing an RR of 3.58 with a 95% confidence interval (CI) of 2.08 - 6.15 (P = 0.0001), when compared to oral ethinyl estradiol users and an RR of 3.04 (95% CI: 1.77 - 5.24; P = 0.0005) when compared to oral norethindrone users.
Age group 35 - 45 years
In females aged 35 - 45 years, VTE risk increased across all groups compared to those in the younger age groups. Oral ethinyl estradiol users (n = 598,925) showed a VTE rate of 0.65% (3,869 VTE events), and norethindrone users (n = 262,663) demonstrated a higher rate of 0.86% (2,251 VTE events). The highest risk was observed in injectable conjugated estrogen users (n = 1,066), with a VTE rate of 2.72% (29 VTE events). Injectable estrogen conferred an RR of 4.21 (95% CI: 2.94 - 6.04; P < 0.0001) compared to oral ethinyl estradiol users and an RR of 3.17 (95% CI: 2.21 - 4.56; P < 0.0001) compared to oral norethindrone users.
| Discussion | ▴Top |
Review of the data from a large inpatient/outpatient administrative database demonstrates that injectable estrogen may be associated with a higher risk of VTE compared to oral ethinyl estradiol and oral norethindrone, as well as an increased risk of VTE compared to those who have not used estrogen and/or progestin. Based on the authors’ literature search, this study is the first to evaluate the associated risk of VTE with injectable estrogen administration.
The data available in the TriNetX Research Network are limited. There is potential selection bias as only the institutions contributing to the database are included. The route of administration in the database is recorded as unknown or not specified for many hormone users, as such, our analysis necessarily will miss some users of estrogens and progestins. We did not account for duration of therapy or other specific VTE risk factors. Whether patients were treated inpatient or outpatient was also not possible to elucidate, as full chart review cannot be performed with a database search. There is a chance that patients were prescribed medication in a setting that did not use the EMR included in the database. However, presumably, these medications would generally be added into the EMR by participating clinicians, so this number of missed prescriptions is assumed to be small. As the database is limited to the search term “injectable,” this term was used as a surrogate measure for “intravenous.” Although it is possible that not all injectable estrogen use was IV, intramuscular and subcutaneous injections of estrogen are not standard clinical practice for HMB treatment, therefore we assumed that the number of non-IV injectable estrogen users was small or negligible. The groups of norethindrone users might include those taking doses other than 5 mg or more of norethindrone acetate, but as these doses are standard clinical practice for HMB treatment, we assumed that the number of patients outside this range were also small. The age range of 10 - 45 was intentionally selected to better capture the population at risk for hormonally induced VTE while minimizing confounders. The lower limit of 10 years was chosen to maximally capture early post-menarchal patients, an important subgroup for whom hormonal therapy is often initiated for HMB. The upper limit of 45 years was chosen to minimize the number of peri-menopausal and menopausal women, who have an inherently higher baseline risk of VTE due to age and may be more likely to use hormonal medication for menopausal symptoms rather than for HMB. Despite these limitations, the absolute number of medical records in the database is large, and presumably all groups were equally subject to the same confounders, as such, the general comparison of groups remains valid. Numbers were too small to accurately stratify VTE risk by dosage of norethindrone, even after excluding the lowest dose of 0.35 mg, which may be important clinically as VTE risk is theorized to be dose-dependent. Lastly, it is not clear why the risk in these data of VTE amongst oral ethinyl estradiol users appears lower than that of norethindrone users or non-hormone users for all age ranges, but we suspect that this reflects prescription patterns. For example, we did not control for underlying risk of VTE or contraindication to estrogen, so it is likely that those prescribed an oral estrogen had a lower baseline risk of VTE. Overall, we suggest that the risk for VTE may be elevated in users of IV estrogen compared to the risk for users of oral ethinyl estradiol and oral norethindrone acetate.
The mechanism of IV estrogen leading to VTE is not well understood. An advantage of IV estrogen treatment is the rapid onset of action, which may be life saving for those with severe hemorrhage. DeVore’s study showed cessation of bleeding within the 8-h study period for most participants. Munro et al found that bleeding stopped in 88% of those taking combined oral hormonal contraceptives and 76% of those taking oral medroxyprogesterone acetate, but with a much longer median time of 3 days [14]. Another option recommended by ACOG for treatment of acute HMB [4] is tranexamic acid, which has a faster onset (5 - 15 min) when administered intravenously [15]. There are, however, no data to suggest that IV estrogen therapy, despite the rapid onset of action, is superior to oral estrogen therapy or progestins for HMB. Authors propose that investigation with a large multicenter detailed record review is needed to accurately determine if IV estrogen carries a significantly higher risk of VTE compared to other hormonal medications for HMB, as this review appears to show, and ask the question: is IV conjugated estrogen a reasonable first line treatment for HMB or, is the risk of VTE so high and the benefit over other hormonal regimens so low that it should be considered in only special circumstances within treatment algorithms? Given that recommendations for IV estrogen are widely based on a single study, and that antifibrinolytic therapy with rapid onset is another potential tool available for treatment of severe HMB, the use of IV estrogen and associated complications would benefit from further study.
| Supplementary Material | ▴Top |
Suppl 1. A detailed list of anticoagulants, including antiplatelets, injectable agents, vitamin K antagonists, and direct oral anticoagulants.
Acknowledgments
The authors report no acknowledgements.
Financial Disclosure
The authors have no financial disclosures to report and this research received no external funding.
Conflict of Interest
The authors report no conflict of interest.
Informed Consent
Not applicable given de-identified nature of dataset.
Author Contributions
Conceptualization: all authors. Methodology: all authors. Data curation and formal analysis: Dr. Citla-Sridhar. Writing, including original draft preparation, reviewing and editing: all authors. TriNetX access: Dr. Citla-Sridhar. All authors have read and agreed to this version of the manuscript.
Data Availability
Data proprietary to TriNetX. The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
ACOG: American College of Obstetrics and Gynecology; AUB: abnormal uterine bleeding; CE: conjugated estrogen; CI: confidence interval; dL: deciliter; EE: ethinyl estradiol; EMR: electronic medical record; g: gram; HMB: heavy menstrual bleeding; ICD: International Classification of Diseases; IV: intravenous; mg: milligram; NE: norethindrone; RR: relative risk; VTE: venous thromboembolism
| References | ▴Top |
- Warner PE, Critchley HO, Lumsden MA, Campbell-Brown M, Douglas A, Murray GD. Menorrhagia I: measured blood loss, clinical features, and outcome in women with heavy periods: a survey with follow-up data. Am J Obstet Gynecol. 2004;190(5):1216-1223.
doi pubmed - Munro MG, Critchley HOD, Fraser IS, Committee FMD. The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions. Int J Gynaecol Obstet. 2018;143(3):393-408.
doi pubmed - Munro MG, Critchley HOD, Fraser IS, Committee FMD. Corrigendum to "The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions" [Int J Gynecol Obstet 143(2018) 393-408.]. Int J Gynaecol Obstet. 2019;144(2):237.
doi pubmed - ACOG committee opinion no. 557: Management of acute abnormal uterine bleeding in nonpregnant reproductive-aged women. Obstet Gynecol. 2013;121(4):891-896.
doi pubmed - Abou-Ismail MY, Citla Sridhar D, Nayak L. Estrogen and thrombosis: A bench to bedside review. Thromb Res. 2020;192:40-51.
doi pubmed - DeVore GR, Owens O, Kase N. Use of intravenous Premarin in the treatment of dysfunctional uterine bleeding—a double-blind randomized control study. Obstet Gynecol. 1982;59(3):285-291.
pubmed - Cockrum RH, Soo J, Ham SA, Cohen KS, Snow SG. Association of progestogens and venous thromboembolism among women of reproductive age. Obstet Gynecol. 2022;140(3):477-487.
doi pubmed - Tans G, Curvers J, Middeldorp S, Thomassen MC, Meijers JC, Prins MH, Bouma BN, et al. A randomized cross-over study on the effects of levonorgestrel- and desogestrel-containing oral contraceptives on the anticoagulant pathways. Thromb Haemost. 2000;84(1):15-21.
pubmed - Harrison RL, McKee PA. Estrogen stimulates von Willebrand factor production by cultured endothelial cells. Blood. 1984;63(3):657-664.
pubmed - Oedingen C, Scholz S, Razum O. Systematic review and meta-analysis of the association of combined oral contraceptives on the risk of venous thromboembolism: The role of the progestogen type and estrogen dose. Thromb Res. 2018;165:68-78.
doi pubmed - Lidegaard O, Lokkegaard E, Svendsen AL, Agger C. Hormonal contraception and risk of venous thromboembolism: national follow-up study. BMJ. 2009;339:b2890.
doi pubmed - Gomes MP, Deitcher SR. Risk of venous thromboembolic disease associated with hormonal contraceptives and hormone replacement therapy: a clinical review. Arch Intern Med. 2004;164(18):1965-1976.
doi pubmed - Bloemenkamp KW, Rosendaal FR, Buller HR, Helmerhorst FM, Colly LP, Vandenbroucke JP. Risk of venous thrombosis with use of current low-dose oral contraceptives is not explained by diagnostic suspicion and referral bias. Arch Intern Med. 1999;159(1):65-70.
doi pubmed - Munro MG, Mainor N, Basu R, Brisinger M, Barreda L. Oral medroxyprogesterone acetate and combination oral contraceptives for acute uterine bleeding: a randomized controlled trial. Obstet Gynecol. 2006;108(4):924-929.
doi pubmed - Pilbrant A, Schannong M, Vessman J. Pharmacokinetics and bioavailability of tranexamic acid. Eur J Clin Pharmacol. 1981;20(1):65-72.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Gynecology and Obstetrics is published by Elmer Press Inc.