| Journal of Clinical Gynecology and Obstetrics, ISSN 1927-1271 print, 1927-128X online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Gynecol Obstet and Elmer Press Inc |
| Journal website https://jcgo.elmerpub.com |
Review
Volume 15, Number 1, March 2026, pages 13-18
Reflections on the Challenges of Infertility-Based Randomized Controlled Trials
Peter Kovacsa, k, Summer Crawfordb, c, Saira Kotharid, Nigel Pereirae, f, Jose Carugnog, Steven R. Lindheimh, i, j
aDunamenti REK, Istenhegyi IVF Center, Budapest, Hungary
bTexas Tech
University Health Science Center Permian Basin, Odessa, TX, USA
cDepartment of
Obstetrics and Gynecology, Texas Tech University Health Science Center Permian Basin, Odessa,
TX, USA
dUniversity of Central Florida/HCA Healthcare Consortium of Greater
Orlando, Orlando, FL, USA
eMount Sinai Fertility, Toronto, ON,
Canada
fDivision of Gynecologic Reproductive Endocrinology and Infertility,
Department of Obstetrics and Gynecology, University of Toronto, Toronto, ON,
Canada
gMinimally Invasive Gynecology Division, Obstetrics, Gynecology and
Reproductive Sciences Department, University of Miami, Coral Gables, FL,
USA
hDepartment of Obstetrics and Gynecology, University of Central Florida,
Orlando, FL, USA
iCenter for Reproductive Medicine, Ren Ji Hospital, School of
Medicine, Shanghai Jiao Tong University, Shanghai, China
jShanghai Key
Laboratory for Assisted Reproduction and Reproductive Genetics, Shanghai,
China
kCorresponding Author: Peter Kovacs, Dunamenti REK, Istenhegyi IVF Center,
Budapest, Hungary
Manuscript submitted October 23, 2025, accepted December 22, 2025, published online January 4,
2026
Short title: Challenges of Infertility-Based Trials
doi:
https://doi.org/10.14740/jcgo1575
| Abstract | ▴Top |
Successful randomized clinical trials (RCTs) are considered the gold standard in clinical research, but require sufficient recruitment, adequate sample size, appropriate participant selection, pre-defined outcomes, and transparent reporting of outcomes. Infertility-related RCTs harbor a unique set of logistical and ethical challenges, as highlighted by our multicenter RCT evaluating clinical differences in low-dose versus high-dose gonadotropin in poor responders undergoing assisted reproductive technology. This is a descriptive review and reflections of frequent difficulties faced by researchers while conducting RCTs and their implications, as well as potential solutions when conducting this type of research. While our RCT in poor responders undergoing in vitro fertilization (IVF) failed to demonstrate superior clinical outcomes with high-dose versus low-dose gonadotropins, the study was significantly underpowered. Despite this, our results help underscore universal difficulties in the recruitment, conduct, and publication of RCTs. We review our challenges and suggest available tools to aid in recruitment and enhance trial completion. Recruitment, conducting, and publishing an RCT have numerous challenges, particularly in an infertile population, which are not unique to our study. Addressing these barriers, despite the shortcomings, provides significant value to the scientific community.
Keywords: Gonadotropin; IVF; Infertility; Randomized controlled trial; Enrollment
| Introduction | ▴Top |
Advancements in healthcare depend on a diversity of research that provides the basis for patient care. Randomized clinical trials (RCTs) represent the most evidence-based research and are considered the gold standard for elucidating significant associations in clinical research due to their unbiased nature, randomization, and control of confounding factors [1, 2]. However, conducting an RCT has become increasingly complex, costly and often fails to meet expected recruitment. Furthermore, infertility-related RCTs harbor a unique set of logistical and ethical challenges specific to this population. Finally, although we recently published data from our RCT, the manuscript was submitted to several peer-reviewed journals with underlying criticisms from each reviewer that included: “underpowered,” “asked the same question as prior studies,” and “an exploratory study.”
Although frustrating, the peer-review process must be endorsed as a learning process. After undergoing multiple edits, the manuscript gained publication [3]. Subsequently, we were encouraged by editors to detail and discuss the challenges in completing our RCT, including when to decide to terminate the trial, and what to do with our findings despite a possible beta (type II) error. Thus, we present the logistic travails of running an RCT, especially an investigator-initiated infertility RCT, and we discuss why it is imperative to publish even negative findings in the case of an underpowered RCT for the purpose of serving future systematic reviews and meta-analyses.
| Our RCT | ▴Top |
“Increased gonadotropin dosing (375 vs. 225 IU/day) does not improve clinical outcomes in poor prognosis patients undergoing IVF: results of a multicenter randomized controlled trial.”
The results of our multicenter RCT aimed to determine whether the use of higher gonadotropin dosing was associated with improved cumulative clinical pregnancy rate (cCPR) in expected poor responders undergoing in vitro fertilization (IVF) [3].
While multiple trials related to this topic have been reported, including several RCTs, various methodological issues have precluded a conclusive approach to these patients [4-8]. According to the latest guidelines, the use of increased gonadotropin dosing is not recommended during IVF; however, often in daily practice, 450 - 600 IU gonadotropins per day are prescribed [9]. In planning this trial, we attempted to design a clinical trial to address issues from the previous trials to better answer the question “is higher dosing better than lower dosing?” This included identification of poor responders based on POSEIDON criteria [10], use of the same stimulation protocols in all arms, the exclusion of the use of oral agents where international unit conversion is not possible, prohibiting dose escalation during treatment cycles, and using internationally agreed clinically relevant endpoints. Furthermore, the trial was designed as a multicenter prospective RCT from six Hungarian IVF centers to enhance external validity.
In determining the necessary sample size, 350 patients in both the lower-dose and higher-dose arms were needed to show the superiority of higher gonadotropin dosing. The trial was initially planned for 1 year based on expected patient enrollment by the co-investigators, with an interim analysis after enrollment of the first 350 patients to assess for the benefits of gonadotropin dosing on clinical pregnancy outcomes. Due to recruitment issues - including a shortage of gonadotropin-releasing hormone (GnRH)-antagonists twice for 2 months, and an unfortunate patient death related to an undisclosed autoimmune disorder that was deemed unrelated to the trial procedures based on the coroner’s exam - the enrollment was extended to 1.5 years. Finally, with no additional resources to maintain the mandated insurance requirement per the Ethics Committee, the joint decision was made amongst the site-investigators to terminate the study prior to meeting our planned sample size.
Overall, of the planned enrollment, 190 participants were randomized, with 98 receiving lower-dose treatment and 92 receiving higher-dose treatment. Analysis of the study’s outcomes revealed the same conclusion of previous RCTs: use of increased dose gonadotropin did not correlate to greater stimulation, embryology, or clinical pregnancy outcomes in expected poor responders undergoing IVF [3]. As a result, rather than a definitive efficacy trial, our RCT was peer-reviewed as an exploratory study, as we failed to show any differences in cCPR in poor responders using either low-dose or high-dose gonadotropins, with a high probability of a type II error.
| Challenges of RCTs | ▴Top |
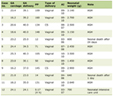
RCTs have become increasingly complex and expensive to conduct. The Consolidated Standards of Reporting Trials (CONSORT) guidelines can serve as a roadmap when the study steps are planned and help identify specific roles for the study, which may vary depending on funding [11] (Table 1). Farrell et al compared the development and execution of an RCT to a business plan, putting forward five basic stages of running a clinical trial. Briefly, these are: 1) initiation, including hypothesis generation and identification of co-investigators; 2) planning, involving preparation of a research plan and finding study personnel at each site; 3) execution, with training of study personnel and subject enrollment; 4) monitoring and control, including follow-up at each site and enrollment of new sites if required; and 5) analysis and reporting [12].
 Click to view |
Table 1. Responsibility Related to Trial
Associated Activities in Pharma-Sponsored, Investigator-Initiated Trials and Our
Own RCT |
Nonetheless, we, as well as many others, failed to meet enrollment goals, which are commonly encountered in RCTs. In a review of a cohort of 114 trials, Campbell et al reported that less than one-third recruited their target participants within their original timeline, and approximately one-third needed an extension to meet the study goals [13].
In retrospect, stage 1 of our “business plan” posited challenges regarding our hypothesis. The lack of universally agreed-upon inclusion criteria has led to difficulty comparing data across existing studies [14]. Two criteria, the Bologna criteria and the POSEIDON classification, have spearheaded enhanced universal identification of poor responders [10, 15]. Despite this, both criteria have faced disapproval among reproductive endocrinologists. The Bologna criteria is a binary classification system that classifies patients as being poor responders or not based on meeting at least two criteria (advanced maternal age ≥ 40 years or other risk factor for poor ovarian reserve, previous evidence of poor ovarian reserve, abnormal ovarian reserve testing), but excludes classifying younger women with abnormal ovarian reserve testing as poor responders [16]. The POSEIDON criteria is a more nuanced stratified classification system that separates expected and unexpected poor responders based on age, ovarian reserve testing, and ovarian response [16]. It includes young women with an unexpected poor ovarian response (POSEIDON 1), whose treatment outcome (pregnancy) may not be different from other normal responders not otherwise classified by POSEIDON. This contrasts with expected poor responders (POSEIDON 3 and 4) [17]. As inclusion criteria such as POSEIDON have been further enhanced, this has led to limitations in recruitment due to the stringent inclusion criteria. While restricting to unexpected poor responders defined as POSEIDON 3 and 4 allowed us to enhance external validity and the generalization of our results, we needed to be more realistic about its impact on study recruitment and consider less stringent criteria for inclusion. Furthermore, as we identified motivated co-investigators, we needed to anticipate and address provider reluctance to prescribe follitropin-δ, which at the time was a new fertility medication potentially limiting their willingness to enroll study patients.
With respect to stages 2, 3, and 4, we lacked dedicated study personnel at each site to assist with execution, monitoring, and follow-up, including enrollment of new sites. Though a collegial relationship was in place with our co-investigators, in hindsight it would have been critical to assess in advance how many subjects a co-investigator realistically was able to enroll based on the characteristics of their patient population and over a defined timeline. In contrast to pharma-sponsored trials, where feasibility questionnaires are often utilized, going by one’s word proved to be inadequate. Considering that IVF services in Hungary are fully reimbursed up to age 45, the incorrect notion among co-investigators was that recruitment would not be an issue.
Unlike investigator-initiated studies, pharma-sponsored studies routinely have investigator meetings detailing the rationale, review of the protocol, and procedures for the study. Furthermore, regular site visits by monitors allow for assessment of enrollment progress and could offer solutions to improve subject recruitment. Successful trial recruitment has been shown to correspond to having a dedicated study nurse [13]. Sully et al reported that involving clinical trial units increased overall recruitment and saved time and costs by assessing efficacy and determining trial progress [18]. In our investigator-initiated clinical trial, despite good intentions with a second online meeting with the goal of re-reviewing the study protocol, this was not enough. Further, updates were not provided at regular intervals, and contact would have provided a constant reminder to enroll patients and to identify barriers that limited subject recruitment. Instead, contact was ad hoc due to a lack of study support personnel. This is particularly imperative when unforeseen events occur, as in our study, which included a medication shortage and an untimely death.
Tools and strategies to aid participant recruitment include local outreach, digital platforms, enhanced patient education, increased amount of time spent with patients answering questions, and utilization of recruitment companies. Further, Paramasivan et al developed the Quanti-Qualitative Appointment Timing (Q-QAT) technique to identify and address challenges with recruitment [19]. Others have used the Quintet Recruitment Intervention (QRI) tool, which helps participants make informed decisions and overall increases RCT recruitment [20]. They both thoroughly assess the consenting process and provide feedback on how to make it more efficient. Utilizing these tools proactively in the early phase of an RCT before recruitment difficulties came to light in future studies should be considered to enhance subject numbers and study results [21].
Difficulties in achieving participant recruitment goals for RCTs can mainly be attributed to under-calculation of financial needs, limited financial resources from grants or pharma support, and improper utilization and spending of financial resources [22]. High costs associated with an RCT’s planning phase are often underestimated. In our study, the only incentive was co-authorship for site investigators, and the study was planned to avoid extra clinic visits without compromising care. This, nonetheless, was not enough to secure adequate enrollment.
| Challenges of Conducting Infertility-Related RCTs | ▴Top |
Infertility-related clinical trials present unique challenges, including methodology, ethical and emotional questions, and financial and operational issues. The sequential stages of fertility treatments (stimulation, egg retrieval, and embryo transfer) create statistical challenges, including what to measure and how to handle patients who undergo multiple treatment attempts within the same study. Live birth is the most important outcome of fertility treatments, but due to long follow-up between treatment and birth, one needs to take into consideration the potential for considerable drop-out rates. Patients who undergo multiple treatment attempts within the same study represent further statistical challenges. For example, if study participants “crossover” from one treatment group to another, particularly if they do not conceive early in the study, it can compromise the study’s randomization and dilute the treatment’s effect. Other methodological issues include the use of a total number of randomized participants versus total number of participants who complete the study. In a 2016 review of 142 IVF RCTs published in 2013 and 2014, approximately one out of three trials reporting live birth rates reported results using a post-randomization denominator, while intention-to-treat analysis (initial total number of randomized participants) should have been used making the data more reliable [23]. This issue is well illustrated by an example regarding different stimulation protocols for ovarian stimulation. Instead of including all participants who initiated treatment, the primary outcome would only be calculated in those who underwent oocyte retrieval or embryo transfer, excluding participants who may have been cancelled for a poor response and never underwent an oocyte retrieval or embryo transfer, thus introducing a bias towards those who responded to a particular stimulation protocol. While this does not represent an intentional manipulation of data, this may occur in infertility studies that may have several different secondary outcomes and treatment milestones, making various combinations of numerators and denominators available to use in data analysis [23]. Additionally, infertility patients frequently pursue complementary and alternative therapies including nutritional supplements, herbal therapies, acupuncture, and ovarian platelet-rich plasma therapy, often concurrently with conventional treatment. This may introduce potential unmeasured confounding factors into fertility research outcomes [24, 25].
Infertility-related studies may also pose ethical and emotional questions including the treatment of vulnerable participants and the potential future child undergoing infertility trials, which requires careful attention of all potential risks. For example, a multiple gestation pregnancy could be desired by the parents but poses significant health risks to both the pregnant woman and the infant(s). Some patients may feel that they are “experimented” with when they undergo their best chance at pregnancy and thus may elect not to participate, while others may agree to anything that promises a successful outcome. Patients undergoing fertility treatment often may feel a mix of hope and desperation, which can compromise their ability to give fully informed consent. Cultural or financial pressures can complicate the decision-making process. While women bear the burden of the treatment, the decision to participate in a trial is shared with the partner or family, thus creating privacy issues. Infertility-related studies are rife with ethical quandaries, including what to do with surplus embryos after a trial, determining fair compensation for participants without exploiting them, and establishing appropriate age limits and inclusion criteria for treatments, which often vary widely based on individual assessment or public funding.
Lastly, regarding financial and operational challenges, recruiting patients is notoriously difficult, and many trials fail to meet their enrollment goals. Reasons include strict eligibility criteria, patient fear of receiving a placebo, and the heavy financial and time burden on participants. Additionally, the financial demands of infertility trials are significant. The high cost of treatment, combined with extensive monitoring and long-term follow-up required to track live births, creates major financial and logistical burdens. In many places, funding is limited, and industry sponsorship has declined, making these trials exceedingly difficult. Finally, clinical trials are subject to an increasing regulatory burden, including extensive paperwork for human subject research. These regulations are designed to protect participants but can significantly increase the cost and time required to launch and complete a trial.
| Reasons for Publishing Underpowered RCTs | ▴Top |
Most journals prioritize significant findings while rejecting studies with negative results, representing a potential for publication bias. We implore to all investigators that underpowered RCTs should be published as they may provide valuable data that can be combined with other studies in systematic reviews and meta-analyses, contributing to the collective body of evidence and influencing clinical practice. Rather than being reluctant to pursue publication, authors should focus on different aspects of their datasets that highlight interesting features of the study population, even if they do not achieve statistical significance based on the primary outcome. Furthermore, such studies may be valuable for demonstrating important findings about the rarity or ubiquity of events, which can be a significant outcome in themselves. Finally, publishing results regardless of outcome, including trials with null results, helps to counteract the publication bias that can skew the perceived body of evidence by masking the true effects of treatments, and it can often serve as pilot studies and provide important information on feasibility, develop realistic estimates of effect sizes, and refine study designs for larger, more powerful trials.
Considerations for authors who want to publish their work include pre-registered protocols that pre-specify analyses and make data publicly available by demonstrating full reporting, regardless of outcome. Investigators should also commit to data-sharing agreements in their protocols to facilitate meta-analyses and individual data pooled analyses, even from under-enrolled trials. Reporting should also emphasize responsible interpretation of the data itself rather than focusing solely on the lack of statistical significance, acknowledging that “underpowered analyses” can be more problematic than “underpowered datasets.” Findings such as ours, even if not statistically significant, can serve to help design future, adequately powered trials by revealing issues with implementation or providing better estimates of effect sizes.
| Conclusions | ▴Top |
Recruitment, conducting, and publishing an RCT have numerous challenges, as highlighted by our own RCT study investigating treatments particularly in an infertile population. These difficulties are not unique to our study. Despite these challenges, RCTs are necessary for advancing clinical care in the field of infertility care. It is critical to address these barriers and to conduct these studies and to encourage publication in order to support clinically applicable advancements in medical treatments, including infertility treatment. Through the lens of our own investigation, strategies to optimize an RCT include the use of a business model approach, implementation of standardized recruitment tools, and utilization of a dedicated clinical team for recruitment and tracking progress. Despite the shortcomings of our RCT, our findings have significant value in lessons learned with respect to study execution and serve to encourage publication to contribute to systematic reviews and meta-analyses in the future.
Acknowledgments
This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the author(s) and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Financial Disclosure
The research did not receive any specific grant from funding agencies in the public, commercial, or non-profit sectors.
Conflict of Interest
The authors declare no conflict of interest that could inappropriately influence or bias this work.
Author Contributions
Peter Kovacs: conceptualization, investigation, writing - review and editing, supervision. Summer Crawford: investigation, writing - original draft preparation. Saira Kothari: investigation, writing - original draft preparation. Nigel Pereira: investigation, writing - review and editing. Jose Carugno: investigation, writing - review and editing. Steven Lindheim: conceptualization, investigation, writing - review and editing, supervision.
Data Availability
Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
Abbreviations
RCT: randomized clinical trial; IVF: in vitro fertilization; cCPR: cumulative clinical pregnancy rate; GnRH: gonadotropin-releasing hormone; CONSORT: Consolidated Standards of Reporting Trials; Q-QAT: Quanti-Qualitative Appointment Timing; QRI: Quintet Recruitment Intervention
| References | ▴Top |
- Murad MH, Asi N, Alsawas M, Alahdab F. New evidence pyramid. Evid
Based Med. 2016;21(4):125-127.
doi pubmed - Burns PB, Rohrich RJ, Chung KC. The levels of evidence and their role
in evidence-based medicine. Plast Reconstr Surg. 2011;128(1):305-310.
doi pubmed - Kovacs P, Nagy DU, Boga P, Zadori J, Sandfeld E, Lindheim SR.
Increased gonadotropin dosing (375 vs 225 IU/day) does not improve clinical outcomes in poor
prognosis patients undergoing IVF: results of a multi-center randomized controlled trial.
Journal of IVF-Worldwide. 2025;3(4):1-13
doi - van Tilborg TC, Eijkemans MJ, Laven JS, Koks CA, de Bruin JP,
Scheffer GJ, van Golde RJ, et al. The OPTIMIST study: optimisation of cost effectiveness through
individualised FSH stimulation dosages for IVF treatment. A randomised controlled trial. BMC
Womens Health. 2012;12:29.
doi pubmed - Berkkanoglu M, Ozgur K. What is the optimum maximal gonadotropin
dosage used in microdose flare-up cycles in poor responders? Fertil Steril.
2010;94(2):662-665.
doi pubmed - Liu X, Li T, Wang B, Xiao X, Liang X, Huang R. Mild stimulation
protocol vs conventional controlled ovarian stimulation protocol in poor ovarian response
patients: a prospective randomized controlled trial. Arch Gynecol Obstet.
2020;301(5):1331-1339.
doi pubmed - Lefebvre J, Antaki R, Kadoch IJ, Dean NL, Sylvestre C, Bissonnette F,
Benoit J, et al. 450 IU versus 600 IU gonadotropin for controlled ovarian stimulation in poor
responders: a randomized controlled trial. Fertil Steril. 2015;104(6):1419-1425.
doi pubmed - Bastu E, Buyru F, Ozsurmeli M, Demiral I, Dogan M, Yeh J. A
randomized, single-blind, prospective trial comparing three different gonadotropin doses with or
without addition of letrozole during ovulation stimulation in patients with poor ovarian
response. Eur J Obstet Gynecol Reprod Biol. 2016;203:30-34.
doi pubmed - Bosch E, Broer S, Griesinger G, Grynberg M, Humaidan P, Kolibianakis
E, Kunicki M, et al. ESHRE guideline: ovarian stimulation for IVF/ICSI. Hum Reprod Open.
2020;2020(2):hoaa009.
doi pubmed - Alviggi C, Andersen CY, Buehler K, Conforti A, De Placido G, Esteves SC, Fischer R, et al. A new more detailed stratification of low responders to ovarian stimulation: from a poor ovarian response to a low prognosis concept. Fertil Steril. 2016;105(6):1452-1453.
- Merkow RP, Kaji AH, Itani KMF. The CONSORT framework. JAMA Surg.
2021;156(9):877-878.
doi pubmed - Farrell B, Kenyon S, Shakur H. Managing clinical trials. Trials.
2010;11:78.
doi pubmed - Campbell MK, Snowdon C, Francis D, Elbourne D, McDonald AM, Knight R,
Entwistle V, et al. Recruitment to randomised trials: strategies for trial enrollment and
participation study. The STEPS study. Health Technol Assess. 2007;11(48).
doi pubmed - Polyzos NP, Devroey P. A systematic review of randomized trials for
the treatment of poor ovarian responders: is there any light at the end of the tunnel? Fertil
Steril. 2011;96(5):1058-1061.e1057.
doi pubmed - Ferraretti AP, La Marca A, Fauser BC, Tarlatzis B, Nargund G,
Gianaroli L, Definition EwgoPOR. ESHRE consensus on the definition of 'poor response' to ovarian
stimulation for in vitro fertilization: the Bologna criteria. Hum Reprod.
2011;26(7):1616-1624.
doi pubmed - Humaidan P, Alviggi C, Fischer R, Esteves SC. The novel POSEIDON
stratification of 'Low prognosis patients in Assisted Reproductive Technology' and its proposed
marker of successful outcome. F1000Res. 2016;5:2911.
doi pubmed - Boots CE, Bernardi LA. Bologna criteria: clinically or academically
relevant? Fertil Steril. 2018;109(1):59-60.
doi pubmed - Sully BG, Julious SA, Nicholl J. A reinvestigation of recruitment to
randomised, controlled, multicenter trials: a review of trials funded by two UK funding
agencies. Trials. 2013;14:166.
doi pubmed - Paramasivan S, Strong S, Wilson C, Campbell B, Blazeby JM, Donovan
JL. A simple technique to identify key recruitment issues in randomised controlled trials: Q-QAT
- Quanti-Qualitative Appointment Timing. Trials. 2015;16:88.
doi pubmed - Donovan JL, Rooshenas L, Jepson M, Elliott D, Wade J, Avery K, Mills
N, et al. Optimising recruitment and informed consent in randomised controlled trials: the
development and implementation of the Quintet Recruitment Intervention (QRI). Trials.
2016;17(1):283.
doi pubmed - Wang R, Chen ZJ, Vuong LN, Legro RS, Mol BW, Wilkinson J. Large
randomized controlled trials in infertility. Fertil Steril. 2020;113(6):1093-1099.
doi pubmed - Griessbach A, Speich B, Amstutz A, Hausheer L, Covino M, Wnfried
Ramirez H, Schandelmaier S, et al. Resource use and costs of investigator-sponsored randomized
clinical trials in Switzerland, Germany, and the United Kingdom: a metaresearch study.
J Clin Epidemiol. 2024;176:111536.
doi pubmed - Wilkinson J, Roberts SA, Showell M, Brison DR, Vail A. No common
denominator: a review of outcome measures in IVF RCTs. Hum Reprod.
2016;31(12):2714-2722.
doi pubmed - Smith JF, Eisenberg ML, Millstein SG, Nachtigall RD, Shindel AW, Wing
H, Cedars M, et al. The use of complementary and alternative fertility treatment in couples
seeking fertility care: data from a prospective cohort in the United States. Fertil Steril.
2010;93(7):2169-2174.
doi pubmed - Sfakianoudis K, Simopoulou M, Nitsos N, Rapani A, Pappas A, Pantou A,
Chronopoulou M, et al. Autologous platelet-rich plasma treatment enables pregnancy for women in
advanced reproductive age with poor ovarian response. Journal of Clinical Medicine.
2019;8(10):1593.
doi
This
article is distributed under the terms of the Creative Commons Attribution 4.0 International
License (CC BY 4.0), which permits unrestricted use, distribution, and reproduction in any
medium, including commercial use, provided the original work is properly
cited.
Journal of Clinical Gynecology and Obstetrics is published by Elmer Press
Inc.